In short: Yellowing of white laundry is caused by the build-up of perspiration residues, limescale and detergent. Most effective solution: sodium percarbonate (active oxygen), 2 tablespoons soaked in hot water for 1 to 4 hours before washing at 60 °C. Avoid bleach on synthetic fibres — it yellows instead of whitening.
At a Glance
Follow the label — the bleaching symbol dictates what is allowed.
Right product — only use what the textile allows.
Thorough rinse — a full cycle limits detergent residues.
Prevention — correct dosage, separate whites, thorough drying.
Whitening by Laundry Type
For yellowed whites, first soak for 2 to 4 hours in percarbonate↗ (1 to 2 tablespoons per litre), then wash at 60 °C if the label allows it.
| Item | Precaution | Programme |
|---|---|---|
| White cotton sheets | Check the bleaching symbol | Cotton programme, normal spin |
| White T-shirts (sweat marks) | Pre-treat locally before washing | Cotton or synthetic programme depending on composition |
| White towels | Thorough rinse and complete drying | Cotton programme, normal spin |
| White underwear | Respect the fragility of elastic | Delicate programme, gentle spin |
| Yellowed net curtains | Fragile fabric, no aggressive bleaching | Delicate programme, minimal spin |
| White linen | Avoid aggressive rubbing | Delicate programme, moderate spin |
Always check your item’s care label before any washing or bleaching.
Why Laundry Turns Yellow
Whites yellow mainly when residues of sebum, detergent and limescale accumulate cycle after cycle, particularly with repeated washes at 30-40 °C.
Yellowing is not a single phenomenon: it is a combination of different chemical reactions depending on the fibre.
Overall yellowing or localised stains?
If you are mainly dealing with yellow underarm stains, also check
our dedicated guide to perspiration stains
. This article covers overall yellowed white laundry, not just a localised area.
Cellulose oxidation (cotton, linen)
Cellulose chains slowly oxidise (air, heat, trace metals), creating yellowish chromophores. This mechanism is documented in classical textile chemistry (Lewin, Textile Research Journal, 1965).
Body residues (sebum, proteins)
Sebum and perspiration residues polymerise then oxidise on skin-contact areas (collar, underarms). If rinsing is insufficient, these deposits become a lasting base for yellowing.
Detergent and mineral deposits
Excess detergent combined with limescale forms a film on the fibre. This film dulls the white, traps soiling and promotes secondary oxidation during storage.
Yellowing induced by certain treatments
Certain finishes or acid treatments can intensify yellow colouring at high heat (Lu & Yang, Textile Research Journal, 1999).
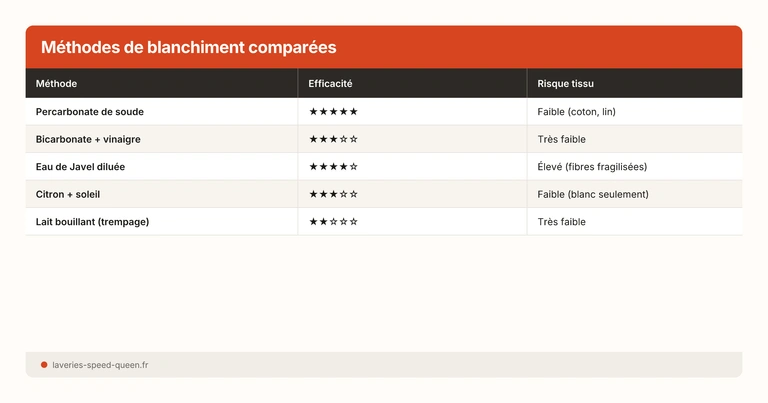
Comparison of Whitening Products
In practice, percarbonate dosed at 1 to 2 tablespoons per litre remains the most effective option for restoring whites, while vinegar (100 to 250 ml) mainly serves to improve rinsing.
Need the full percarbonate guide?
Here, we focus on yellowed laundry. If you are looking for how percarbonate works, at what temperature it becomes truly useful, what it cannot do and the safety precautions, also read
sodium percarbonate and laundry: temperature, safety, real uses
.
| Product | Practical dosage | Effectiveness on yellowing | Suitable textiles |
|---|---|---|---|
| Sodium percarbonate | 1 to 2 tbsp/L for soaking (hot water) | High for oxidative and organic yellowing | Cotton, linen, resistant whites (if label allows oxygen-based bleach) |
| Hydrogen peroxide (H2O2) | 3%: local application or diluted bath | Good as targeted pre-treatment | Fragile white textiles compatible with oxygen bleach |
| Baking soda | 1 to 2 tbsp per cycle | Low to medium (wash support, not a primary whitener) | Almost all textiles, useful against odours and hard water |
| White vinegar | 100 to 250 ml at rinse stage | Indirect (dissolves mineral deposits, improves rinsing) | Cotton, synthetics, towels; caution on sensitive fibres |
Quick read: to “restore whites”, percarbonate and hydrogen peroxide are the main active agents. Baking soda and vinegar are mainly maintenance auxiliaries.
Detergent residues
Too much detergent or insufficient rinsing leaves residue on the fibres. These residues oxidise over time and turn yellow. This is the most common cause.
Hard water
Hard water limescale deposits on the fibres and creates a greyish or yellowish film. The harder the water, the faster the phenomenon.
Perspiration and sebum
Mineral salts from sweat and sebum (skin oil) react with the fabric and cause yellowing, especially at the underarms, collar and cuffs. See our article on stubborn stains for sweat marks.
Prolonged storage
White laundry stored for months in a cupboard (especially if not washed before storage) yellows through natural fibre oxidation in contact with stagnant air.
Curative Solutions
For heavily yellowed laundry, the most reliable method is an oxygen soak of 2 to 4 hours followed by a full cycle at 60 °C with an extended rinse.
Respect the bleaching symbols
The triangle symbol on the label indicates what is allowed. Empty triangle: all bleaching.
Triangle with two lines: oxygen bleach only (no chlorine bleach).
Crossed-out triangle: no bleaching at all. Never use a product that is not compatible with the textile.
Percarbonate vs Bleach: Which to Choose?
The choice between these two whitening agents depends on the textile, the severity of yellowing and what the label allows. Here is a detailed comparison to help you choose.
| Criterion | Sodium percarbonate | Bleach (sodium hypochlorite) |
|---|---|---|
| Whitening power | High (active oxygen) | Very high (active chlorine) |
| Fibre aggressiveness | Moderate — gentle on cotton and linen | High — weakens fibres over time |
| Compatible textiles | Cotton, linen, white synthetics (if label allows oxygen-based bleach) | White cotton only (empty triangle on label) |
| Synthetics (polyester, nylon) | Compatible | Not allowed — bleach yellows synthetics |
| Environmental impact | Low — breaks down into water, oxygen and soda ash | High — releases chlorine, toxic to aquatic environments |
| Safety | Good — no irritating fumes | Moderate — chlorine fumes, never mix with vinegar |
| Typical dosage | 1-2 tbsp / litre for soaking | Per manufacturer, max 30 min soak |
| Activation temperature | 40 °C minimum to be effective | Works at any temperature |
Verdict: sodium percarbonate is the best choice in 90% of cases — effective, versatile, gentle on fibres and the environment. Reserve bleach for extreme cases (heavily yellowed white cotton, stubborn marks) and only if the empty triangle symbol appears on the label.
Three Families of Whitening Agents
Sodium percarbonate (active oxygen)
Oxygen-based whitening agent, effective and less aggressive than bleach. It releases active oxygen on contact with water, breaking down the molecules responsible for yellowing. Suitable for cotton, linen and most resistant white textiles. Avoid on silk, wool and delicate fabrics.
Hydrogen peroxide
Gentle whitener, often used as a localised pre-treatment on sweat marks or yellowed areas. Works similarly to percarbonate but in liquid form. Useful for textiles that tolerate oxygen bleach but not chlorine bleach. Always test on a hidden area first.
Bleach (sodium hypochlorite)
The most powerful, but also the most aggressive. Only allowed if the empty triangle symbol appears on the label. Bleach weakens fibres over time and yellows certain synthetics (polyester, nylon). Follow the manufacturer's dosage and rinse thoroughly. Reserve it for cases where active oxygen is not enough.
The Soaking Technique
Extended soak before washing
For heavily yellowed laundry, soaking in a basin with a whitening agent (percarbonate or hydrogen peroxide depending on the label) before machine washing allows the product to work in depth. Follow the dosage and duration recommended by the product manufacturer. Then rinse in the machine with a full cycle. This step is particularly effective on cotton sheets and white towels.
Careful Re-washing (Without Whitening Agent)
Sometimes a good wash is enough
If the yellowing comes from detergent residues, re-washing with a full cycle and a thorough rinse can solve the problem. Wash whites separately to avoid any colour transfer. At the laundromat, professional machines offer particularly effective rinsing thanks to their water volume (50-60 L).
The Sun Method: Natural UV Whitening
Ultraviolet (UV) rays from the sun have a natural whitening effect on cellulosic fibres (cotton, linen). This phenomenon, known as photobleaching, has been used for centuries — long before the invention of chemical whiteners.
How to Proceed
- Wash the laundry normally (with or without a whitening soak depending on the severity of yellowing).
- Hang it out still wet on a line or flat on a clean surface, in full sunlight. Residual moisture activates the whitening effect of UV — dry laundry does not whiten in the sun.
- Leave exposed for 2 to 4 hours, turning once halfway through for an even result.
- Re-dampen if necessary: if the laundry dries before the end of exposure, spray with water to maintain the effect.
Limitations of This Method
- The effect is gradual: it can take 3 to 5 sessions for heavily yellowed laundry.
- Only works well on whites — the sun fades coloured textiles.
- Ineffective indoors (windows block a large proportion of UV).
- For maximum results, combine percarbonate soak + sun drying: the percarbonate works in depth, the sun finishes the job on the surface.
The sun does not replace washing
Sun drying whitens fibres, but does not remove dirt or bacteria. Always wash the laundry before exposing it. For optimal drying, alternate between sun (whitening effect) and tumble dryer (speed, softness).
What Does NOT Work (or Makes Things Worse)
- Ignoring the label — unauthorised bleaching can destroy fibres or permanently dull the fabric.
- Overdosing detergent — excess detergent leaves residue on the fibres, which oxidises and contributes to yellowing. Less detergent = better rinsing.
- Mixing whites and colours — even a garment that "doesn't bleed" can transfer micro-pigments over multiple washes.
- Using bleach on coloured cotton — bleach discolours irreversibly. Even a white T-shirt with a coloured print will be damaged.
- Mixing bleach and vinegar — this combination triggers a chemical reaction that releases chlorine fumes, dangerous for the respiratory system. Never mix these two products.
- Soaking too long in bleach — beyond 30 minutes, bleach starts weakening cotton fibres. The laundry becomes fragile and develops holes after the next few washes.
- Sun drying without rinsing after treatment — if you sun dry laundry still loaded with whitening product, you risk water marks and uneven discolouration spots.
- Using hot water on untreated perspiration stains — heat sets sweat proteins into the fibres and makes yellowing permanent. Always pre-treat sweat marks in cold water before washing.
Preventing Yellowing: The 4 Pillars
Prevention is better than cure. Yellowing of white laundry is a gradual process — by adopting good habits now, you will avoid catch-up soaking later.
1. Wash regularly at 60 °C (not only at 30 °C)
Why always washing at 30 °C eventually dulls whites: at low temperatures, body oils, some surfactants and part of the biofilms are less effectively dissolved. The laundry looks clean visually, but residual layers accumulate cycle after cycle.
2. Dose the detergent correctly
Overdosing detergent is one of the primary causes of yellowing: surfactant residues trapped in the fibres oxidise over time and turn yellow. Follow the manufacturer’s instructions, adjust for your water hardness, and prefer slight under-dosing to over-dosing.
3. Dry quickly and completely
Laundry that stays damp for hours (forgotten in the machine, poorly spun, dried in a poorly ventilated room) yellows faster than laundry dried quickly. Tumble drying or air drying in full sunlight is ideal.
4. Store clean, dry and aired
Never store laundry that is still damp or unwashed. Natural fibre oxidation in contact with stagnant air in a closed cupboard is a common cause of yellowing in long-stored laundry.
| Laundry type | Regular cycle | Hot maintenance cycle |
|---|---|---|
| Everyday white T-shirts | 30-40 °C | 60 °C every 3 to 5 washes |
| White sheets and pillowcases | 40 °C | 60 °C at least every 2 washes |
| White towels | 40-60 °C | 60 °C almost every time if textile allows |
| Delicate white textiles | 30 °C delicate | No hot cycle; use occasional active oxygen instead |
Dose the detergent correctly — too much detergent leaves residues.
Wash whites separately — colour transfer dulls whites. See our temperature guide.
Sufficient rinsing — a full cycle limits deposits.
Dry quickly — laundry left damp for a long time yellows faster. Find our tips in the drying guide.
Store clean and dry — never store laundry that is still damp or unwashed.
As an Amazon Partner, we earn a small commission on purchases made via the affiliate links in this article — at no extra cost to you. This helps us maintain this site and produce free guides.
Restore the brightness of your white laundry. Our laundromats in Blagnac and Croix-Daurade offer large-capacity machines and effective rinsing. See our prices.
Methodology and Sources
- The active oxygen mechanism is documented by the detergent industry’s technical resources: sodium percarbonate (Na2CO3·1.5H2O2) releases hydrogen peroxide in aqueous solution, which oxidises the chromophores responsible for yellowing (AISE - International Association for Soaps, Detergents and Maintenance Products (lien externe)).
- The interpretation of the triangle symbol (bleaching allowed, oxygen-based only, or prohibited) follows the GINETEX / ISO 3758 (lien externe) standard.
Sources and References
- Washing temperature guide
- Stubborn stains and pre-treatment guide
- Machine detergent dosage: how much to use
- Sodium percarbonate: complete guide
- Laundry drying guide
- White vinegar and laundry: uses and limits
- Perspiration stains and yellow marks
- Textile care symbols (GINETEX / ISO 3758) (lien externe)
- AISE - International Association for Soaps, Detergents and Maintenance Products (lien externe)
- Lewin M., Textile Research Journal (1965), cellulose oxidation mechanism