In a nutshell: soda crystals (sodium carbonate, pH 11) are a detergent booster in hard water and a powerful degreaser for heavily soiled laundry. Dosage: 1-2 tablespoons per load. Gloves required. Never use on wool, silk or aluminum. Don’t confuse with baking soda (gentler), sodium percarbonate (bleaching agent) or caustic soda (dangerous).
Soda crystals ≠ caustic soda
Never confuse soda crystals (sodium carbonate, Na2CO3, pH 11) with caustic soda (sodium hydroxide, NaOH, pH 14). Caustic soda is a corrosive product that causes severe burns. It has no use on laundry. Soda crystals are irritant but not corrosive — the difference is fundamental.
At a glance
Sommaire
- At a glance
- Soda crystals: what are they?
- The big comparison table: three products not to confuse
- The real uses of soda crystals on laundry
- Dosage: the practical chart
- Precautions: what to take seriously
- What you must NOT use soda crystals on
- Detailed instructions
- Soda crystals in homemade detergent: what to know
- What doesn’t work (or works poorly)
- Storage and preservation
- When to prefer another product
- Sources and references
Not baking soda — pH 11 vs pH 8.3: soda crystals are far more alkaline and aggressive.
Gloves required — pH 11 irritates skin and eyes. Always handle with gloves.
Booster, not substitute — soda crystals soften hard water and degrease, but don't replace detergent.
1-2 tablespoons per load — no more. Alkaline overdosing damages fibers.
Never on wool, silk, aluminum — strong alkalinity destroys protein fibers and attacks certain metals.
Soda crystals: what are they?
Soda crystals↗, or sodium carbonate (Na2CO3), are an alkaline mineral salt. In aqueous solution, they produce a high pH (11-11.5) giving them degreasing and water-softening properties.
It’s an age-old product — used for centuries in laundry and household cleaning. It’s found as a component in many industrial detergents, where it acts as a “builder” agent: it neutralizes water hardness (calcium) to allow detergent surfactants to work better.
Where do they come from?
Soda crystals come either from natural natron deposits (Africa, North America) or from the Solvay process which manufactures them from salt and limestone. The end product is the same: anhydrous or hydrated sodium carbonate (with 1 or 10 water molecules depending on the commercial form).
The big comparison table: three products not to confuse
The confusion between soda crystals, baking soda and sodium percarbonate↗ is the most common source of errors in “natural cleaning” content. These are three chemically distinct products with different uses.
| Criterion | Soda crystals | Baking soda | Sodium percarbonate |
|---|---|---|---|
| Chemical name | Sodium carbonate | Sodium bicarbonate | Sodium percarbonate |
| Formula | Na2CO3 | NaHCO3 | Na2CO3·1.5 H2O2 |
| pH in solution | 11 - 11.5 | 8.3 | 10.5 |
| Main action | Degreaser, water softener | Deodorizer, light supplement | Oxygen-based bleaching |
| Strength | Strong | Mild | Medium-strong |
| Protection | Gloves required | None needed | Gloves recommended |
| On wool/silk | Prohibited | Compatible (low dosage) | Not recommended |
| On colors | OK (normal dosage) | OK | Discoloration risk |
| Temperature | 40 °C minimum | Any temperature | 40-60 °C minimum |
| Indicative price | 3-5 €/kg | 3-5 €/kg | 5-8 €/kg |
For dedicated guides to each product: baking soda and laundry and sodium percarbonate and laundry.
The real uses of soda crystals on laundry
Soften hard water
This is their most logical use. Sodium carbonate neutralizes limescale (Ca2+ and Mg2+ ions) by converting it to insoluble calcium carbonate. Result: detergent works better because it's no longer 'captured' by limescale. Especially useful if your water exceeds 25 °f.
Boost detergent in hard water
1 tablespoon in the drum is enough to improve your detergent's effectiveness in hard water. You can even slightly reduce the detergent dosage (by 10-20%) when adding soda crystals.
Degrease heavily soiled laundry
Work clothes, kitchen towels and aprons soaked in grease respond well to a soda crystal soak. The alkalinity saponifies fats (a chemical reaction that turns grease into soluble soap).
Soak heavily grimy laundry
For heavily grimy laundry (mop cloths, cleaning rags, kitchen towels), a 2-3 hour soak in hot water (50 °C) with 2-3 tablespoons of crystals per liter strips effectively before machine washing.
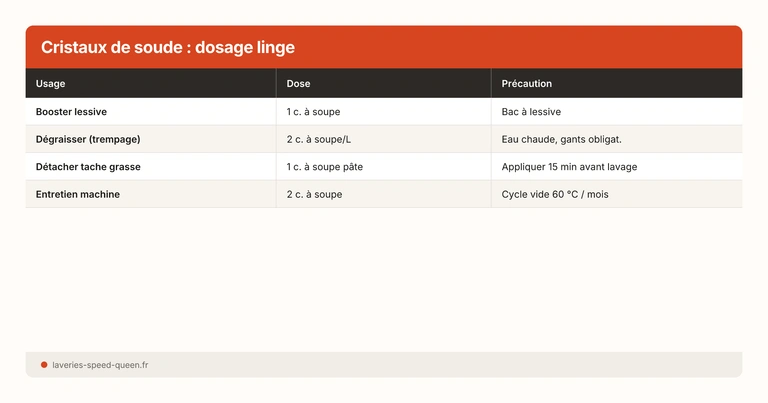
Dosage: the practical chart
Overdosing is the most common mistake with soda crystals. More is not better — excess alkalinity can damage fibers, leave whitish residue and irritate skin when wearing the clothes.
| Use | Dosage | Temperature | Instructions |
|---|---|---|---|
| Boost detergent (hard water) | 1 tbsp (15 g) | 40-60 °C | Directly in the drum with the laundry |
| Heavily soiled / greasy | 2 tbsp (30 g) | 40-60 °C | In the drum, with your regular detergent |
| Soaking (grimy laundry) | 2-3 tbsp / liter | 50-60 °C | Basin, soak 2-3 hours, then machine wash |
| Machine maintenance (empty cycle) | 2-3 tbsp | 60 °C | Empty drum, full cycle |
1 tablespoon = approximately 15 g
Soda crystals come as powder or small crystals. A level tablespoon equals about 15 grams. If using a measuring cup, 15 g of soda crystals occupy about 20 ml (crystals are less dense than water). See our detergent dosage guide to optimize your entire wash protocol.
Precautions: what to take seriously
Soda crystals are not a harmless product. Their pH of 11 puts them among the strong alkalines in household products. This is not comparable to baking soda (pH 8.3) which can be handled bare-handed without risk.
Gloves required — pH 11 irritates skin within minutes of contact. Rubber or nitrile household gloves are sufficient.
Avoid eye splashes — in case of eye contact, rinse thoroughly with clean water for 15 minutes. Consult a doctor if irritation persists.
Don't inhale the powder — fine powder can irritate the airways. Handle in a ventilated area, without creating a dust cloud.
Keep away from children — the white powder looks like sugar or flour. Label the container clearly and store out of reach.
Don't use on skin — unlike baking soda, soda crystals are not suitable for cosmetic or body use.
What you must NOT use soda crystals on
- Wool — wool keratin is degraded by strong alkalines. Fibers lose their softness, felt and become brittle. For wool, prefer baking soda (pH 8.3) or a special wool detergent.
- Silk — silk fibroin reacts the same way as keratin to high pH. The fabric dulls, weakens and loses its sheen irreversibly.
- Aluminum — soda crystals attack aluminum through a chemical reaction (forming sodium aluminate). Don't wash laundry with aluminum elements (buttons, rivets, zippers).
- Specially finished textiles — waterproof, water-repellent (Gore-Tex type) or UV-treated fabrics lose their finish under strong alkalinity.
- Lacquered or varnished surfaces — if you wash furniture covers with lacquered elements or decorative textiles with finishes, alkalinity can attack the finish.
Detailed instructions
Method 1: boost detergent in hard water
If your water is hard (over 25 °f), half your detergent is “wasted” fighting limescale instead of cleaning laundry. Soda crystals neutralize this limescale, allowing detergent to focus on cleaning.
- Pour 1 tablespoon of soda crystals directly into the drum, with the laundry (not in the detergent drawer — crystals can clog the conduits).
- Add your detergent to the drawer, reducing the dose by 10-20% from your usual amount.
- Start the cycle at 40 °C minimum. Crystals dissolve better in warm water.
This method has a double advantage: your laundry is better washed and you save detergent.
Method 2: soaking heavily soiled laundry
For greasy kitchen towels, oil-soaked work clothes or towels that have become rough and smelly.
- Fill a basin or tub with hot water (50-60 °C).
- Add 2-3 tablespoons per liter of water. Stir well with a utensil (not bare-handed — gloves).
- Submerge the laundry and soak for 2-3 hours. For heavily grimy items, overnight soaking is possible.
- Rinse, then machine wash with your regular detergent. The soak stripped the grease — the machine finishes the job. For more on soaking, see our pre-wash and soaking guide.
Method 3: machine maintenance
Soda crystals can also maintain the washing machine itself. Limescale builds up on the heating element and pipes — soda crystals dissolve it.
- Pour 2-3 tablespoons into the empty drum.
- Run a cycle at 60 °C, cotton program, empty drum.
- Repeat once a month if your water is very hard.
This complements descaling and detergent drawer cleaning.
Soda crystals in homemade detergent: what to know
Soda crystals are a common ingredient in homemade detergent recipes. They act as water softener and degreaser. But their use in homemade detergent has the same constraints as using them alone:
- Not on wool or silk — any homemade detergent with soda crystals is incompatible with these fibers.
- Minimum temperature — a homemade detergent with soda crystals doesn’t perform well in cold water.
- Controlled dosage — the total pH of the homemade detergent must remain manageable. Excess soda crystals makes the mix too alkaline.
Soda crystals are not a detergent
This is a classic trap in “zero waste” cleaning recipes. Soda crystals soften water and degrease, but they have no surfactants — the detergent component that lifts dirt particles from fabric and keeps them suspended in rinse water. Without surfactants, soiling redeposits onto the laundry. For a complete detergent, you also need a soap (Marseille, for example) that acts as the surfactant.
What doesn’t work (or works poorly)
- Mixing soda crystals and vinegar — acid-base reaction that neutralizes both. You get sodium acetate (neutral salt) and water. Zero cleaning power.
- Using in cold water — crystals dissolve poorly in cold water. Whitish residue on laundry and very limited degreasing action.
- Overdosing thinking 'more = better' — excess alkalinity damages fibers, leaves residue and can irritate skin when wearing.
- Using them as a bleach — soda crystals don't bleach. They have no active oxygen like sodium percarbonate. If you want to whiten, percarbonate is what you need.
- Using on all textiles — wool, silk, aluminum, special finishes: the list of contraindications is real.
Storage and preservation
Soda crystals are stable in open air — unlike sodium percarbonate which degrades with humidity. But a few rules are good practice:
- Sealed container: prevent the powder from absorbing moisture, which causes it to clump.
- Clear labeling: the white powder looks like other products (baking soda, percarbonate, sugar, salt). A labeled container avoids dangerous mix-ups.
- Out of reach of children and pets: pH 11 is irritant. Store high up or in a locked cabinet.
- Shelf life: unlimited if kept dry. Soda crystals don’t lose their properties over time.
When to prefer another product
Soda crystals aren’t the right answer for every situation.
- To whiten: use sodium percarbonate (active oxygen).
- To deodorize: use baking soda (gentler, no textile risk).
- To soften laundry: soda crystals soften water, not laundry. For feel, see our fabric softener guide.
- To remove stains: depending on the stain, a targeted treatment is better. See our tough stains guide.
- To disinfect: soda crystals have no significant disinfecting action. See our guide on disinfecting laundry.
As an Amazon Partner, we earn a small commission on purchases made via the affiliate links in this article — at no extra cost to you. This helps us maintain this site and produce free guides.
For heavily soiled laundry or large volumes of kitchen towels and work clothes, our professional machines offer 60 °C programs ideal for soda crystals’ action. Find our laundromats at Blagnac, Croix-Daurade and Montaudran. Payment CB sans contact ou espèces. Check our pricing.
Sources and references
- Baking soda and laundry: the real uses
- Sodium percarbonate and laundry: temperature, safety, real uses
- Homemade detergent: recipes, limits and real risks
- Detergent dosage guide
- Descaling a washing machine
- Pre-wash and soaking: when and how
- White vinegar and laundry: uses and limits
- Washing kitchen towels
- Sodium carbonate (Na2CO3) — alkaline salt, pH 11-11.5 in solution, “builder” agent in industrial detergents
- Solvay process — industrial synthesis of sodium carbonate from salt (NaCl) and limestone (CaCO3)